Part Five of a Series on Soap. Part One, On the Quest for Soap. Part Two, What is Soap? Part Three, On the Quest for Lye. Part Four, A Homemade Soap Recipe are linked if you’re interested.
Making soap is inherently chemistry. Sure, it’s relatively easy chemistry, but it’s chemistry none-the-less.
Back in high school chemistry they taught us about ‘limiting reactants.’ I always remember the bike analogy. Imagine you have two bike frames and six bike tires: how many bikes can you build? Two bikes. Each bike needs two tires and a frame so you’ll run out of frames before you run out of tires. If you only have two bike frames, it doesn’t matter if you have one hundred bike tires because you can’t build any more bikes.
My first batch of soap had zero measuring or math involved. I put some oil in a Pyrex measuring cup, mixed up some lye (meaning I just eyeballed it), and dumped the two together. What resulted was soap, and I was really excited, but then things didn’t work out as well as I’d hoped. I poured the liquid mixture into some molds and let them sit. According to everything I’d read, the soap should harden over a few days and then you can remove the bars from the molds. Even after a week and a half I couldn’t remove the soap. It was hard on top but still liquid underneath. It was like each soap bar was a model for the Earth’s plate tectonics. The top layer would slide and drift over the liquid layer and any cracks would leave a volcano of beige-white soap-lava oozing out of the soap fault line. They’d eventually solidify into a soap mountain range until I messed with the bars again. New fault lines, new soap volcanoes, and the soap-mantle remained ever so fluid.
Eventually I just grabbed the whole glob, liquid and solid, of each bar and smashed them into balls. They were very oily when I hand-molded them, and it was pretty obvious what the problem was: I used way too much oil/not enough lye. Back to the shitty bike analogy, the lye I used was the bike frames and the oil was the tires. Way too many tires to make bikes out of. So the tires (oil) just kinda hung out in the garage in a massive pile along with relatively few finished bikes (soap).
This did have the nice side effect of the soap being very gentle. It didn’t dry my skin out and left my hair feeling amazing after washing with it. It also lathered really well. It felt nice to use. As for texture and consistency, no bueno, the soap would crumple with the smallest force applied to it. It’s just not a very durable soap.
So how do you know how much oil and lye to add together to make as many bikes as possible? Luckily people have already figured that out. You just have to do a little math.
Saponification Value
I stumbled upon a Wikipedia article about this thing called saponification value and it was exactly what I needed to find even if I didn’t know I needed to find it. It basically gives the value of lye you must add to oil to completely saponify it. You just measure your oil, do a little math, and the resulting number is the amount of lye you need to add. Easy!
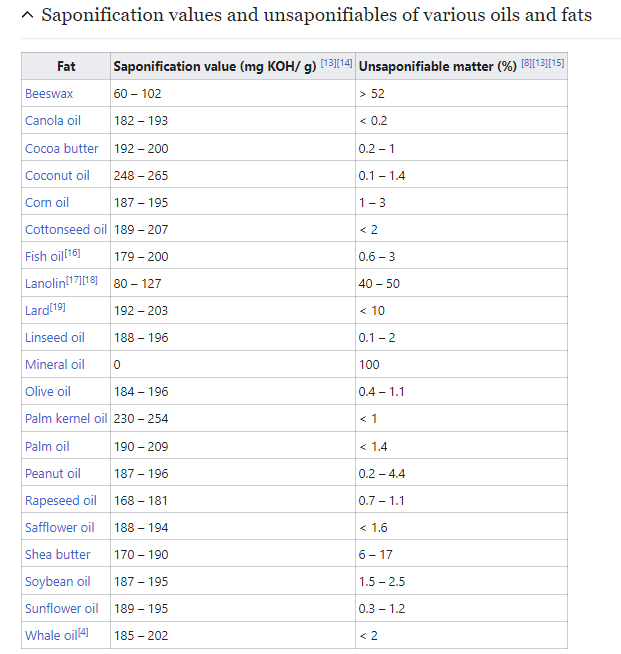
The values given in the Wikipedia table are labeled as milligrams per gram. They’re also the values for using potassium hydroxide (caustic potash) and not sodium hydroxide! Oil requires less lye to saponify than KOH (due to a lower molecular mass…I think…) and luckily the page notes the conversion factor: 1.4. Divide these values by 1.4 and that’s your number for using NaOH instead of KOH.
Obviously you should use a scale if you’re really going to get into soap-making, even occasionally. I’d recommend everyone have a nice kitchen scale because, well, they’re handy. Even if you don’t have a use for it now it’s one of those things that is periodically needed. They’re like $20 so just go buy one!
A Cheap, Quick, and Easy Recipe Revisited
Referencing my last post with the cheap, simple, easy-mode soap recipe: I used 300 g of canola oil. Why? Because canola oil is cheap and you might already have a bunch in your kitchen. Why faff around using fancy, expensive, and unique oils when you can just use the trash you already have on hand? Using canola oil isn’t a rule, and once again feel free to experiment. You can use any oil you want (almost. note that mineral oil can’t be saponified; it’s a different oil altogether and won’t work), just check the saponification value and do the math.
As for the amount of lye we’re going to use the saponification value for canola oil: 190 mg/g. Firstly, divide by 1.4 because we’re using NaOH and not KOH, and this gives a value of 135 mg/g. We’re using 300 g of oil, so 300*135 is 40,500 mg of lye. Since there are 1,000 mg in a g, just move that decimal over and realize you need about 40 g of lye. I’m rounding these numbers down because of reasons…
You want all the lye to react with the oil. Lye isn’t really nice to wash your hands with (all of that stuff about it being caustic and eating flesh/metal), and as stated before an oil-heavy soap lathers better and is gentler on your skin. In short you do not want to use all the lye you need, because a little unreacted oil is good to have. Apparently pro soap makers call this ‘superfatting’ and this is another tweakable you can do with your soaps.
I’ve been using 90% lye based on the numbers and this seems to work nicely even if the soap is still a bit harsh. 40 g is the upmost limit you should use, and you can go much lower than 90% if you want. The soap will become softer and gentler to use, but this is a tradeoff that probably becomes undoable past a certain point because the soap will become soft and brittle. I haven’t found this point yet (I’m still experimenting…) so I’m going with 90% lye for a ‘safe’ soap.

So 90%*40g is 36g. That’s the amount of lye you’ll use. The best thing about saponification values and knowing how they work is that they allow you to really make your own batches instead of blindly relying on a recipe. Sure recipes work but what if you don’t want to make a ton of soap? What if you just want to give it a try before you make five pounds of soap and screw it all up? Or what if you do want to make a ton of soap? Saponification values and somewhat knowing the math and science behind the process lets you scale up or down your recipe and tweak whatever you want. You can play around with the formula, make some 60% lye soap and see what happens! It might be a failure, oh well, and then you can tweak in the other direction next time.
Check out my YouTube channel about off-grid green energy setups!
Or my Instagram where I post pointless artistic pics and shitty poems every whenever I get around to it.
Or my other blog where I sometimes post stories.

Leave a comment